Noninvasive Urine Test for Early Detection of Cervical Cancer Virus Proteins
10/7/2024
New urine-based test shows promise in making cervical cancer screening more accessible and less invasive
Cervical cancer is the fourth most common cancer in women. The current cancer screening methods are mostly invasive and some of them involve detection of human papillomavirus (HPV) DNA. However, recent advances in the field suggest the detection of HPV16 E7 oncoproteins as a more clinically relevant method for screening cervical cancer. Now, researchers have developed a noninvasive alternative method to detect HPV oncoproteins in urine samples that could revolutionize cervical cancer screening.
Cervical cancer is one of the most common cancers in women, with approximately 660,000 new cases and 350,000 deaths worldwide in 2022 alone. Almost all cases are linked to high-risk human papillomavirus (HPV) infections. Current screening methods involve detecting HPV DNA, but emerging research suggests that measuring the cancer-causing activity of HPV may provide a more accurate assessment of cancer risk.
Can a new, super-sensitive test accurately measure proteins linked to HPV in urine to help detect cervical cancer?
A group of researchers led by Professor Etsuro Ito from the Department of Biology, Waseda University, Japan, along with Professor Toshiyuki Sasagawa from Kanazawa Medical University, Japan, and Dr. Martin Müller from the German Cancer Research Center, Germany investigated to develop an ultrasensitive enzyme-linked immunosorbent assay (ELISA) to detect high-risk HPV16 E7 oncoproteins in urine. Their findings were published in the Microorganisms journal on 14 June 2024.
Ito explains, “Cancer can be prevented by vaccination before it develops and by regular screening. But screening is a big hurdle for young women.” He further adds, “Our new urine test can detect HPV16 E7 proteins, which are critical markers of cervical cancer risk, at extremely low levels. This means that women may be able to screen for cervical cancer without the discomfort and inconvenience of a traditional Pap test.”
Current screening methods for cervical cancer typically involve a Pap smear or an HPV DNA test, both of which require a visit to a healthcare provider and can be uncomfortable for many women. This new urine test offers a noninvasive alternative, which could encourage more women to participate in regular screening.
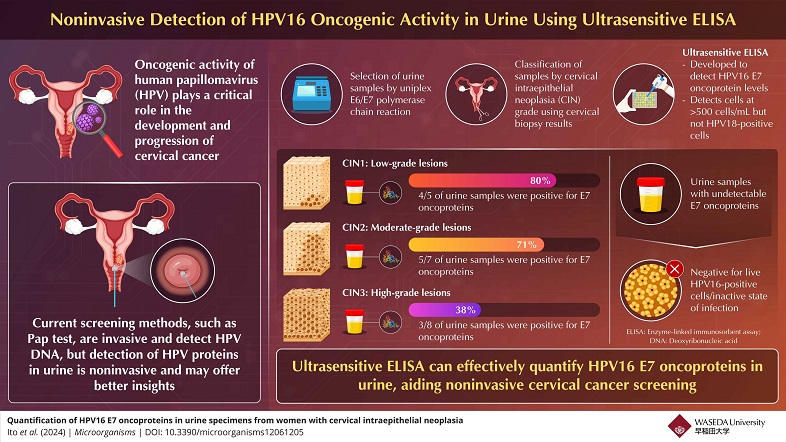
The researchers used ELISA to detect the E7 oncoproteins in urine samples. The test was able to identify these proteins in the urine of women with different stages of cervical intraepithelial neoplasia (CIN), a precursor to cervical cancer. The ELISA test detected E7 proteins in 80% of women with CIN1, 71% with CIN2, and 38% with CIN3, suggesting that the presence of E7 oncoproteins correlates with lower-grade CIN lesions. The researchers theorize that this discrepancy may be due to variations in the HPV life cycle or oncogenic activity.
It says, “We believe that the E7 oncoprotein is critical in the early stages of HPV-related cervical carcinogenesis and E7 may play a more significant role in the progression of CIN1 and CIN2 than in CIN3.”
This innovative approach aligns with global health goals to reduce cervical cancer rates, especially in low- and middle-income countries where access to traditional screening methods is limited. With further development and validation, this urine test could become a standard tool in the fight against cervical cancer, helping to save lives through earlier detection and treatment. The development of a noninvasive urine test for detecting HPV-related proteins represents a significant step forward in cervical cancer screening. It offers a promising solution to increase screening rates and reduce the incidence of cervical cancer worldwide.
It concludes, “This new method holds great promise for early detection and prevention of cervical cancer. We are optimistic that further development and validation of this assay will lead to its widespread use in clinical settings.”
